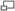Reversible Processes
An analysis of thermodynamically reversible processes, also discussing the work done in a reversible process
Overview
Definitions
Working substance (WS) = the WS is used as the carrier of heat energy. The state of the WS is defined by the values of its properties, e.g. pressure, volume, temperature, internal energy, enthalpy. These properties are also sometimes called functions of state.
<br/>
Key facts
A thermodynamic process is reversible if it may be reversed by some infinitely small change in the conditions affecting it.
Because of the conditions which it must satisfy, no practical process can be considered thermodynamically reversible.
The work done in a reversible process, following a change in volume from  to
to  , is given by:
, is given by:
 where
where  is the pressure.
is the pressure.
A thermodynamic process is reversible if it may be reversed by some infinitely small change in the conditions affecting it. When reversed, it will pass back through each stage it assumed in the direct sense, and it will have the same state at each point in the direct and reverse process. Therefore, an equal amount of work will be absorbed in the reverse process as is given out in the direct process, and also an equal amount of heat will be rejected in the reverse process as was supplied during the direct process.
In order to be reversible, a thermodynamic process must satisfy certain conditions. For example, a thermodynamic process can not be reversible if:
- there is heat transfer between bodies of different temperatures;
- there is friction between moving parts, since friction always represents a loss;
- there are intermolecular forces;
- there are eddies or turbulence in the working substance (WS), since these are irreversible.
Work Done In A Reversible Process
Imagine that the WS is enclosed in a frictionless cylinder with piston, and is initially characterized by the volume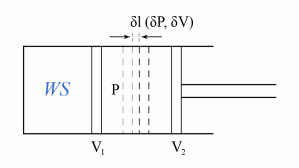
 Login
Login